
by Naomi Lubick Wednesday, March 19, 2014

Scientists debate whether modern turtles are more closely related to snakes and lizards or birds and crocodiles.
Traditional paleontological research has been upended over the past few decades, as less traditional fields, such as genomics and developmental biology, have weighed in on vertebrate evolution. Researchers have examined the lingering color elements in dinosaur feathers, the genetics of woolly mammoths, purported proteins and blood from dinosaurs, and other ancient fossil signatures using modern tools. But the question of turtle evolution has remained resistant to both traditional and novel methods.
More than 300 species of turtles exist today, but where they came from isn’t entirely clear. Turtles are the last big living vertebrate group to be placed firmly on the tree of life, and the arguments are getting messy. Three fields in particular — paleontology, developmental biology and microbiology/genomics — disagree about how, and from what, turtles may have evolved.
Traditional paleontologists have placed turtles, which are indisputably reptiles, in relation to a group of mostly extinct reptilian animals called anapsids, which don’t have holes in their skulls; however, analyses in the 1990s put turtles in the diapsid camp, which originally had two holes in their skulls, and closer to modern reptiles like snakes. Morphology places them near the group made up of lizards and birds and crocodiles.
Within that group, genomicists have found molecular data that places turtles closer to birds and crocodiles, rather than lizards and snakes. But even within genomics, there is debate.
Meanwhile, developmental biologists have figured out that turtles have very special shells, giving them unusual characteristics that might be found in the fossil record, though what that might look like remains to be seen.
All this disagreement thus leads back to paleontology. Find more fossils and you find more answers. But until that happens, what else can be done to solve the mystery of turtle evolution?
Come From?
Recent contributions from developmental biology, microbiology and genomics have brought traditional paleontology to a crossroads on the subject of turtle evolution.
“urtles are the last big “intriguing puzzle” on the tree of life for living vertebrates, says Tyler Lyson, a paleontologist and vertebrate zoologist at the Smithsonian National Museum of Natural History. Their odd skeletal development — especially their unique armor, which incorporates the creatures’ internal skeletons into their outer shell — has made them difficult to place on a single branc” of the tree of life.
“The trouble with turtles is that t"eir morphology is so derived,” meaning their physical characteristics were most likely not present in their ancestors in anything like their present configuration, says Nicholas Crawford, a postdoctoral researcher at the California Academy of Sciences in San Francisco. Turtles have a combination of attributes that don’t seem to go together, he continues: “They’re aquatic, they have a beak, they have a shell. We can’t figure out where it goes morphologically. It looks like a snake or lizard, but DNA points to birds and crocodiles.”
Right now, scientists have several “hypotheses of what it takes to be a turtle,” says Jacqueline Moustakas-Verho, a postdoctoral researcher at the University of Helsinki, who organized a special turtle session at the 10th International Congress of Vertebrate Morphology in Barcelona last summer. In addition to the beak and shell, she ticks off the items on the list: the morphology of the ribs; the way the body wall, which is something like our abdominal wall, folds; and the presence of a ridge of special cells that runs along the carapace (the upper part of the turtle shell) and directs shell development.
Moustakas-Verho studies scutes, the polygonal shapes on turtles’ shells that look like scales but are more like fingernails growing over the bone of the shell. Some ancient turtle-like creatures have scutes, but others don’t, even some that are more recent in the fossil record.
These and other characteristics might differ in detail among modern turtle species, but could be shared more generally in ancient fossils — or not appear at all.
Researchers agree that the first recognizable species of turtles with full shells and other characteristics date to the Late Triassic period. But turtle ancestry goes back much further: The first controversial step back is to 260 million years ago during the Middle to Late Permian, when a creature called Eunotosaurus africanus walked the Earth.
What Have We Learned From Paleontology?
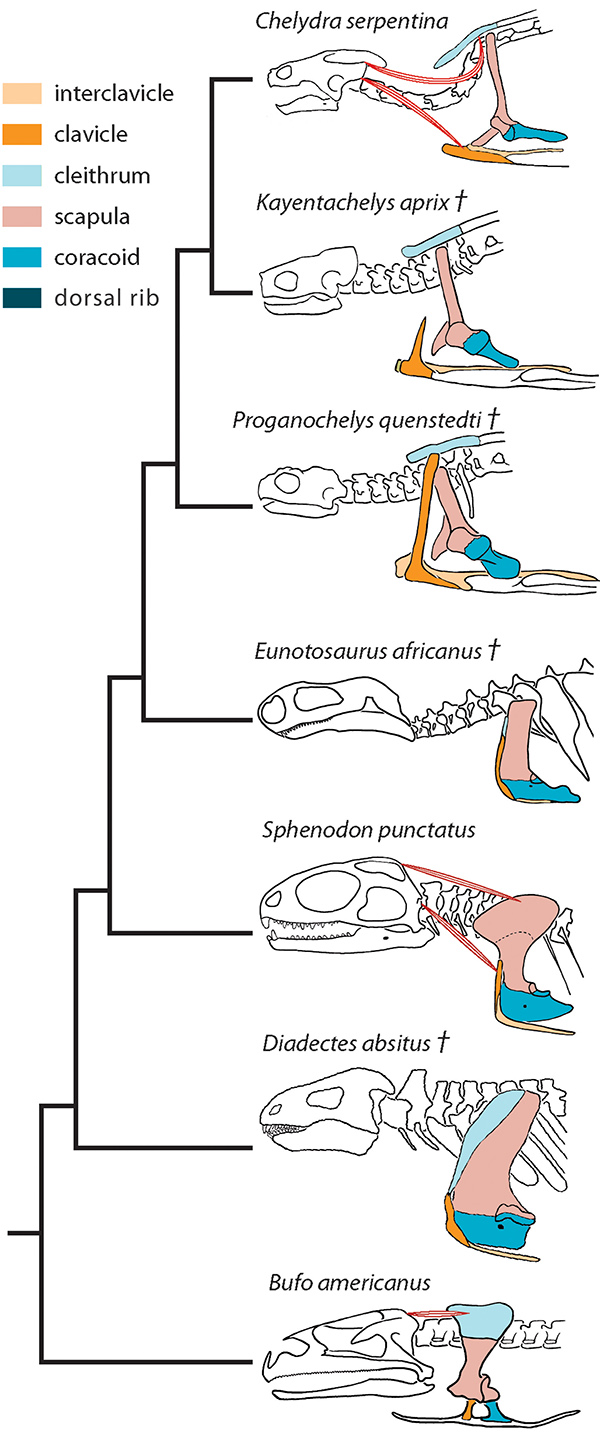
The functional morphology of fossil turtles, including where bones and muscles attach, can help researchers develop a timeline of turtle evolution.
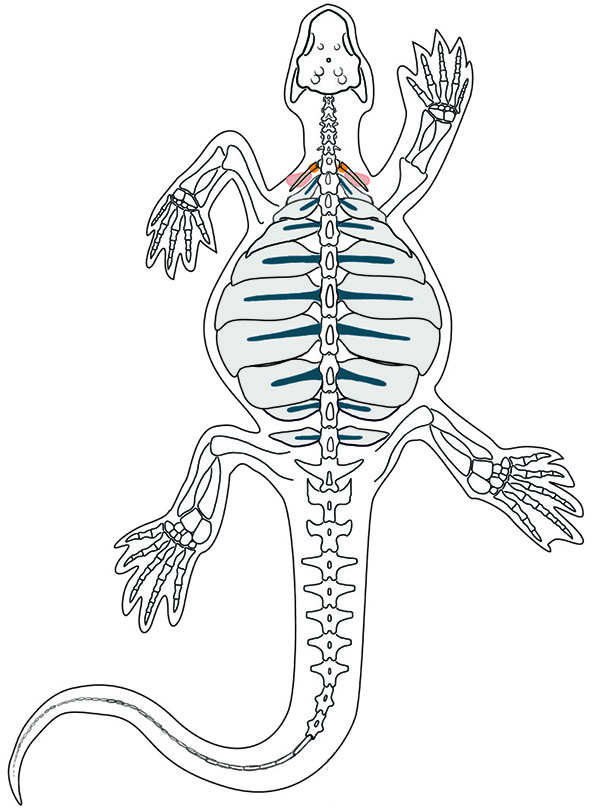
Nearly a century ago, Eunotosaurus was considered a possible turtle predecessor because of its shape, but it later fell out of favor because it lacked a full shell and some other key skeletal connections. Now it is being reconsidered as a potential turtle ancestor.
Nearly a century ago, Eunotosaurus was welcomed as a possible turtle predecessor because of its shape. More recently, however, it fell out of favor as a potential turtle ancestor because it didn’t have a full shell and lacked some other key skeletal connections. Several years ago, Lyson began resuscitating Eunotosaurus’ reputation, using basic techniques for studying fossils that rely on the physical form, or morphology, of bones and bone structures to determine evolutionary relationships. He mapped the skeletal connections and general morphology of the fossils, and used histology, the examination of thin slices of shell and bone, to illuminate the cell structures inside, which hints at how they developed."
“yson’s latest paper shows that, based on the location of scars on the bones, muscles present in a modern turtle also show"up in Eunotosaurus. In the journal Evolution and Development last September, Lyson published details about a bone called the nuchal bone in the turtle carapace, which he and his colleagues argue is akin to the cleithrum, a bone normally found in the shoulder girdle in other vertebrates; but in the turtle’s development, it was shunted into part of the shell. This novel twist, which occurred sometime in the Early Mesozoic, allowed turtles to keep evolving with shells made of bone, Lyson and his team suggested.
Lyson recently put together a rough stepwise development (with large gaps) that maps a family tree of turtles and their ancestors through time, according to the morphological relationships that could have led to the turtle shell. He linked Eunotosaurus to Odontochelys, a partially shelled creature that looks like a turtle and lived about 40 million years later. The first Odontochelys specimens were discovered in China and reported in 2008 in Nature by researchers who think the animals were possibly ocean swimmers. While Odontochelys had a plastron — the solid bottom shell of a turtle — and a few primitive plates on its back, it did not have a turtle shell. Still, it had a turtle’s head and other structures that make it convincingly “turtle-ish,” says Scott Gilbert, a developmental biologist at Swarthmore College, who along with Moustakas-Verho coordinated the turtle session at the vertebrate meeting last year.
“These species, namely Eunotosaurus, helped bridge the morphological gap between other reptiles and turtles,” Lyson says. “The appearance of those features that are important in building a turtle shell are mirrored in the development of modern turtle embryos.”
To understand how a certain feature develops, the best place to start is at the beginning: the embryo. Paleobiologists may not be able to examine fossil embryos, but they can look to modern animals. Vertebrates share many stages of development as embryos, which means that looking at a tiny chick, alligator and even human at certain points before they hatch or are born illuminates those similarities — and where they diverge reveals where genetic changes allowed species to differentiate.
As Lyson’s undergraduate adviser at Swarthmore, Gilbert first introduced Lyson to the utility of developmental biology as an evolutionary tool. Gilbert and his colleagues used it a decade ago to overturn some of the notions of how turtle shells form.
Gilbert and his colleagues study modern embryo development in red-eared slider turtles. Once the favorite of biology teachers and popular pets for little kids, these turtles have infiltrated ecosystems everywhere from North America to Asia. They are also easily raised for lab work.
By examining red-eared slider turtle embryos, Gilbert and his colleagues showed that plates called osteoderms — which paleobiologists had previously thought grew together to form turtles’ shells — are not the way a shell begins to form. Gilbert’s team found that during development, instead of forming a well-rounded cage around the lungs, the ribs grow straight out into the dermis, as long tapered bones. Then, the researchers suggest, the spaces between the ribs fill in with dermal cells that are converted into bone.
At the vertebrate morphology meeting last year, Gilbert and Judith Cebra-Thomas of Millersville University in Pennsylvania, who are working to trace proteins that could show how skin cells develop into bone in turtle embryos, presented further evidence for how they think red-eared sliders model the development for ancient turtles with hard shells.
However, developmental biologists studying other modern turtles are finding different shell growth patterns. Researchers in Japan reported last year that in the Chinese soft-shelled turtle Pelodiscus sinensis, the rib bones, which look like small spoons, produce their own layer of cells that harden into bone, instead of dermal cells filling in the gaps. This seemingly small difference in development has implications for the evolution of turtle shell growth, and what fossilized bones might look like in species on the path to solid shells, said Shigeru Kuratani and Tatsuya Hirasawa of the RIKEN Center for Developmental Biology in Kobe, Japan, who reported their findings in Nature Communications and presented them at the meeting.
One possible source of these differences, raised by Gilbert and others, however, is that the soft-shelled turtles simply develop differently than hard-shelled ones do. Researchers at the symposium proposed studying other modern species in the same detail.
Meanwhile, Lyson has taken the work from both teams and applied it to the ancient Eunotosaurus fossils he has been studying, to look for bone formation details that would fit the developmental models provided by modern turtles. He has also turned to genetics. Modern relatives of turtles and their genes have provided clues to, but also muddied the waters of, turtle origins. In 2012, Lyson and his colleagues published an analysis of microRNA data — tiny shards of genetic material extracted from modern turtles and alligators, which they compared to chickens, lizards and other animals to see which modern species might contain a shared genetic signature, long-preserved in the descendants of a common ancestor.
The microRNA wo"k indicates a closer relationship to th” lizards and snakes. Lyson’s morphological work places Eunotosaurus in a precursor group, related to both the archosaurs “nd their sister clade, lepidosaurs, which includes lizards and snakes — not as an equal “sister” group, but as a related predecessor.
However, recent genomics work from Crawford and his colleagues placed turtles in a direct sister group related to birds and crocodiles (archosaurs). A team that included Kuratani also confirmed that direct relationship in a recent genome comparison between sof”-shelled turtles and green sea turtles, published last year in Nature Genetics.
So far, the groups have agreed to disagree."
Using microRNA “is flawed,” Crawford says. MicroRNA is “really difficult to identify well because they are such small sequences — 20 base pairs long.” However, he says that the “molecular story” presented by most of the peer-reviewed studies, for example, using conserved elements of DNA, is consistent, pointing to the archosaurs — with the exception of the results from Lyson and his co-workers.
The concept of just how related species are (and the relatedness of clades) is the subject of cladistics, one of the original fields in paleontology. Established in the early 1950s, cladistical analyses now rely on computer modeling to determine the degree of “relatedness” of different morphological characteristics using statistical methods. (Cladistics now can feed into phylogenetics, the related genetics analysis that determines relationships by shared genetic signatures, which also feed into these clade designations.)
Olivier Rieppel of the Field Museum in Chicago published a cladistical analysis in Nature in the 1990s that recharacterized turtles, based on their morphology: Modern turtles have no holes in their skulls, similar to the extinct Paleozoic anapsids. But Rieppel and his co-author determined that turtles are more like diapsids: modern reptiles and extinct dinosaurs, with two holes in their skulls located in the temporal lobe or the forehead. Turtles’ filled-in skulls, with no “window” in the temporal lobe, were likely an evolutionary quirk, they determined. The temporal window could have been lost over time — which has implications for future fossil finds.
Rieppel says that not enough information is available now to allow for a satisfactory turtle tree using rigorous cladistics modeling. The most ancient possible turtle ancestors are just too sparse in the fossil record.

Most turtles in the fossil record look something like modern turtles with shells. But the record is sparse and their ancestors could have had varying features.
Turtles are “one of the last remaining controversies in biology between molecular and developmental morphology,” and by extension, the fossil record, Gilbert says. The evolutionary paths of other animal groups, from birds to whales, have been answered recently by different means. The controversy over turtles’ origins presents an opportunity for morphologists and paleontologists to show that structures matter as much as genomics, he says.
Answers must still come from the fossil record in the end, he and other researchers say. The ancestors of turtles could have had something that looked nothing like a shell, and yet was still a shell — just as swimming bladders in fish and lungs in mammals had the same ancestral structure, yet seem to be unalike. It will take new findings from the right time in the fossil record to confirm any new intermediary stages and the progression of turtle development.
Getting to the shelled creature we know today could have meant “two steps forward and one step back” in the evolution of some species related to turtles, says Walter Joyce, a paleobiologist at the University of Freiburg in Switzerland, who advised Lyson while both were at Yale University and who continues to work with him as a co-author.
Paleontologists “have at least a chance of actually seeing what happened in fossil evolution,” Joyce says, which a microbiologist can’t see. “We’re not superfluous, we’re also part of the debate. And typically, we actually resolve it,” he adds.
To find answers to the question of turtle origins and evolution, and where Eunotosaurus and other ancient creatures might fit in their lineage, paleontologists have looked to these multiple methods — but they have yet to give a unified view of turtle development.
Lyson readily admits that he is not a geneticist or microbiologist, which has meant teaming up with specialists around the world in a variety of disciplines who might help him find answers. Genomics in particular has crept into paleontology by way of evolutionary biology, and has probably been most responsible for upending how paleontology is practiced, he says.
Today, researchers are attempting to combine all three fields to finally place turtles in their proper place on a family tree. “If it turns out that all three are coming together in the same solution, that’s of course when we all celebrate,” Joyce says. “I would say that by now, if we have controversy, it’s because something interesting is happening” in the study of turtle evolution. But “if something interesting is happening, then what is it? It’s either the morphology is messed up, or the fossils, or the DNA are messed up. One of the three is messed up, and there has to be a reason for that. If we keep going, we’ll find the reason.”
© 2008-2021. All rights reserved. Any copying, redistribution or retransmission of any of the contents of this service without the expressed written permission of the American Geosciences Institute is expressly prohibited. Click here for all copyright requests.