
by Mary Caperton Morton Tuesday, June 5, 2018

Credit: K. Cantner, AGI.
Lead is one of the most ubiquitous heavy metals in nature, and, for decades, it was a common ingredient in a wide range of consumer goods. After numerous studies detailed its toxicity and concluded there is no safe amount of lead in the human body — especially in children — new laws and regulations curbed its use dramatically. And yet, lead still appears in many products, particularly cosmetics, traditional medicines and folk remedies that are often lightly regulated.
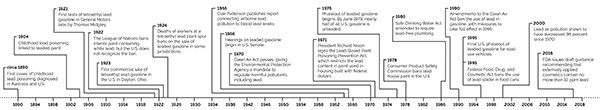
Just a few decades ago, when leaded gasoline still flowed from gas pumps and lead paint brightened the walls of many homes, lead poisoning occurred across all socioeconomic classes in the U.S. The federal government banned the use of leaded paint in houses in 1978, and followed with a ban on the sale of leaded gasoline for on-road vehicles in the 1990s. Research conducted by the Centers for Disease Control and Prevention (CDC) found that, in the late 1970s, more than 77 percent of the U.S. population had elevated blood lead levels — some high enough to cause irreversible brain damage and death. But a follow-up study showed that, by 1994, the percentage of people with elevated blood lead levels had fallen to just 4.4 percent — a striking drop resulting from one of the most successful public health campaigns ever waged in the U.S. But the scourge of lead from other sources remains.
Credit: K. Cantner, AGI.
“We’ve made tremendous progress in this country against the traditional sources of lead,” says Philip Landrigan, a pediatrician and epidemiologist at the Icahn School of Medicine at Mount Sinai in New York City. But today, according to the CDC, more than half a million children in the U.S. are still affected by high blood lead levels, often from exposure to decades-old peeling paint, lead-polluted soils, or what scientists classify as “atypical sources,” a group that includes cosmetics and folk remedies.
To continue making progress against lead poisoning, doctors and scientists need to determine where the metal is still slipping through the cracks into our everyday lives. Studying unique cases, or “sentinel events,” in which individuals or small groups of people have been affected by lead from atypical sources, sheds light on how and where people are still coming into contact with lead with the hope of curtailing further exposure to these same sources.

Leaded gasoline was developed in the 1920s to improve performance and reduce knocking in combustion engines. In the U.S., it began to be phased out in the 1970s due to the toxicity of tetraethyl lead. Credit: above: David Brodbeck, CC BY 2.0.
Lead is toxic to people of all ages, but children are especially vulnerable, says Alan Woolf, a pediatrician and toxicologist at Boston Children’s Hospital. “Children are more susceptible to lead poisoning because of their curious nature and tendency to put everything in their mouths,” Woolf says. But their susceptibility also relates to physiology: “Kids absorb more lead through the gut than adults, and their immature kidneys cannot excrete lead as efficiently as adults,” he says.
The primary pathway for lead intoxication is ingestion; lead does not readily enter the body through the skin. Once absorbed through the gut into the bloodstream, lead makes its way around the body where it tends to be mistaken for calcium because lead and calcium fit into the same cellular receptors. As “Trojan” calcium, lead moves into cells where it can wreak havoc. “Many cellular functions are calcium-dependent,” Woolf says. Instead of facilitating metabolic reactions, lead torpedoes normal cellular functioning, sometimes killing the cell. When lead invades cells in target organs such as the liver, kidneys and brain, the fallout can lead to permanent tissue damage, organ failure and death.
Blood lead levels are used to assess how much lead is circulating in a person’s bloodstream. The CDC recommends medical intervention for levels above 5 micrograms per deciliter (µg/dl). Medical interventions begin with identifying and removing the source of lead; acute cases — when blood lead levels rise above 45 µg/dl — warrant chelation therapy, in which chelating agents that bind lead and help clear it from the body are introduced into the bloodstream.
Left untreated, lead can make its way into storage reservoirs in the body. In adults, as much as 90 percent of residual lead is stored in bone tissue. Children tend to store less lead in their bones, but as children grow and their bones are remodeled, stored lead can be released back into the bloodstream, triggering new episodes of elevated blood levels and cellular malfunction.

A 1919 advertisement for Dutch Boy "White-Lead" paint touted the product's "durability and resistance to weather." Credit: Don O'Brien, CC BY 2.0.
The earliest diagnosed cases of childhood lead poisoning emerged in the late 19th century. In 1892, for example, doctors at the Brisbane Children’s Hospital in Australia noted that 10 children initially misdiagnosed with meningitis turned out to have acute lead poisoning. The lead was traced to paint in the children’s homes, and the product was subsequently banned in Australia in 1914. That year also saw one of the first recorded cases of childhood lead poisoning in the U.S., documented in a case study published in the American Journal of Diseases of Children. Decades passed, however, before doctors realized the severity of long-term effects from lead poisoning.
“When colleagues and I first started working on lead poisoning in the late 1960s, the view at that time was that high doses of lead could cause acute poisoning, which made children horribly sick,” Landrigan says. “We used to see children who had eaten paint chips in comas, in convulsions. Many died; and the ones that survived were left with obvious brain damage.” As devastating as acute lead poisoning can be, the long-term effects of lead exposure are equally sobering: Lead is a neurotoxin that targets both the central and peripheral nervous systems, capable of causing permanent brain damage even in low doses.

Peeling paint has long been a major pathway to lead exposure in the U.S. and elsewhere, even after leaded house paint was banned. Credit: ©iStockphoto.com/XiFotos.
“Over the last few decades, we’ve learned that lead is the worst kind of gift that keeps on giving,” Landrigan says. Children exposed to even low doses of lead tend to exhibit reduced IQ, shortened attention spans, disruptive behaviors, dyslexia and other learning disabilities. “When we track their long-term development, we find that these kids are more likely to drop out of school and have run-ins with the law in adulthood,” he says. “We now know from studies done on low-dose lead toxicity that even small doses of lead can be very harmful, especially to young children and unborn children in the womb.”
“Heavy metals like lead, arsenic, mercury, aluminum, zinc, chromium and iron are found in a wide variety of cosmetics and personal care products, including lipstick and other lip products, whitening toothpaste, eyeliner, nail color, foundations, sunscreens, eye shadows, blush, concealer, moisturizers and even eye drops,” says Janet Nudelman, director of the Campaign for Safe Cosmetics organized by the nonprofit Breast Cancer Prevention Partners. But if ingested lead is so toxic, why does it appear in products like lipstick? “Some metals are intentionally added as ingredients, while others are contaminants of the raw materials or are present in the colorant,” Nudelman says.

Lead-tin-yellow, a yellow pigment composed of lead tin oxides, was a popular choice among artists of old, including 17th-century Dutch painter Johannes Vermeer, who used it in "The Milkmaid." Credit: public domain.
“Like asbestos, lead has certain properties that make it attractive to people and manufacturers,” despite its known hazards to public health, Landrigan says. Lead has been mixed into pigments since antiquity because it reacts with elements such as oxygen, sulfur, manganese and iron to produce chemical salts with brilliant colors. For example, lead oxide was long used in both interior and exterior house paints because of its bright white tone and resistance to weathering.
In 2012, scientists at the U.S. Food and Drug Administration (FDA) published a study in the Journal of the Society of Cosmetic Chemists in which they tested more than 400 lipsticks sold in the U.S. for total lead content. The researchers found that almost all of the lipsticks tested contained small amounts of lead, with an average lead concentration of 1.1 parts per million (ppm). Just over a dozen of them had lead levels of about 3 ppm, and the highest concentration observed was 7.2 ppm.
The lipstick study revealed that, in most cases, the lead was not intentionally added to the lipstick but instead was naturally present in trace amounts in some ingredients. “Based on our surveys, we determined that manufacturers are capable of limiting lead content in cosmetic products to 10 ppm or less if they are careful about selecting their ingredients and follow good manufacturing practices,” the FDA team wrote in the study. As lead is mainly absorbed through the digestive tract, not through the skin, lead exposure from lipstick and other topical cosmetics is minimal, the researchers noted, reporting that they “determined that exposure to 10 ppm lead from incidental ingestion of cosmetic lip products is very small and cannot be measured in routine blood testing.”

In a 2012 study by the U.S. Food and Drug Administration (FDA), researchers tested 400 lipsticks commercially available in the U.S. and found lead concentrations ranging from near zero up to 7.2 parts per million (ppm), with an average of 1.1 ppm. A draft guidance to the cosmetics industry issued by FDA recommends that externally applied cosmetics contain no more than 10 ppm. Credit: ©iStockphoto.com/TheCrimsonMonkey.
The FDA issued draft guidance to the cosmetics industry in 2016 recommending that externally applied cosmetics, including lipstick, should contain no more than 10 ppm lead. On its website, the agency notes that “most cosmetics on the market in the United States generally already contain less than 10 ppm of lead,” but that “some cosmetics from other countries contain lead at higher levels.”
In a 2016 study published in Environmental Science & Technology, scientists tested lipsticks commercially available in China and found lead concentrations up to 10,185 ppm. The average concentration was 497 ppm, with lower-priced products tending to contain more lead. “A woman may ingest 1.8 kilograms of lipstick inadvertently over a lifetime,” the team wrote, “which represents a significant exposure pathway depending on [lead] concentration in lip products.”
Traditional products and folk remedies, which are often produced with untested ingredients and distributed without labels or ingredient lists, can be even more perilous than commercial products, particularly for children.
“A lot of these folk products are imported informally, through families, often by well-meaning grandparents who are trying to pass on their native cultural traditions to their children and grandchildren,” says Suzette Morman, a geochemist with the U.S. Geological Survey in Denver, Colo. “These remedies are perceived as healthy, and not at all hazardous, but they’re generally not regulated and they can turn out to be nightmares from a toxicology standpoint.”

A man applies eyeliner around the eyes of a boy in Kolkata, India. Known variously as kohl, kajal, al-kahal, surma, tiro, tozali and kwalli, such eyeliners are sometimes composed of crushed galena, or lead sulfide, which is toxic, although less bioavailable than some other forms, such as lead oxide or lead carbonate. Credit: ©Shutterstock.com/clicksabhi.
When a child or adult presents with high blood lead levels, atypical sources like traditional medicines and folk remedies must be considered once common sources, such as paint, occupational exposure and industrial pollution, are eliminated. “If a child comes in with an elevated blood lead level and they are living in an apartment that was built in 2016, and nobody in the family works in lead-related occupations, we then have to play detective and delve into other sources of lead that might not be so routine,” Woolf says.
In 2011, Woolf and a team of doctors from Boston Children’s Hospital — one of the leading treatment centers for heavy metal poisoning in the U.S. — published a case study involving a six-month-old boy of Nigerian descent. The boy — who arrived to a well-child physical with eyeliner on his eyelids — appeared healthy, but a routine blood test revealed a blood lead level of 13 µg/dl, more than twice the CDC’s recommended action limit.
When the child’s case came to the attention of Woolf and his colleagues, they suspected the eyeliner could be to blame for the lead in the boy’s system. A form of eyeliner traditionally used in parts of the Middle East and Africa is so notorious for its high lead concentrations that it cannot be legally imported into the U.S. Known in different places as kohl, kajal, al-kahal, surma, tiro, tozali or kwalli, the eyeliner is used cosmetically to enhance the eyes, even of very young children, and is also thought by some cultures to ward off evil spirits. Unfortunately, the material is often composed of galena, the mineral form of lead sulfide.
Lead displays different degrees of bioavailability — a measure of how readily a substance makes its way into the bloodstream and throughout the body — so hazards from lead-containing products often depend on the type of lead involved.

Kohl is applied in many cultures to enhance aesthetic appearance, or as a medicine purported to strengthen eyes and protect against disease or evil. Credit: Ako Mahmoodi, CC BY 3.0.
“When we looked at the eyeliner under a scanning electron microscope, we found it was almost pure galena,” says Geoffrey Plumlee, a geochemist with the U.S. Geological Survey in Reston, Va., who, along with Morman, has collaborated with Woolf and his colleagues on several atypical lead poisoning cases. “Of all the lead minerals, galena is one of the least bioaccessible. That explained why the infant’s blood lead levels were [elevated] but not screaming high.”
Further investigation revealed that the eyeliner had been purchased from a street vendor in the Nigerian city of Ilorin, where it’s called tiro, and imported to the U.S. by the infant’s grandparents. The parents had applied the eyeliner to the infant’s eyelids three to four times a week since he was 2 weeks old to “improve attractiveness and promote visual development,” Woolf’s team wrote in the report, published by the CDC.

Galena crystals are often characterized by their cubic habit, or shape, and distinct cleavage planes at both macroscopic and microscopic scales. Scanning electron microscopy (inset) confirmed that the tiro eyeliner applied to a Nigerian boy and analyzed in a 2012 study was composed largely of tiny galena crystals (field of view is about 0.1 millimeters). Credit: James St. John, CC BY 2.0; inset: U.S. Geological Survey, Crustal Geophysics and Geochemistry Science Center.
Lead sulfide is not readily absorbed through the skin or the mucus membranes, Woolf says. But as young children often rub their eyes and put their fingers in their mouths, the lead from the tiro likely entered the infant’s bloodstream through ingestion, he says, although some could have also been absorbed through the conjunctival surfaces of the eyes or from ingested tears.
After applications of the tiro on the infant ceased, his blood lead levels gradually dropped over a matter of months, and the child never showed outward signs of illness. The case led Woolf’s team to reach out to the Nigeria Center for Disease Control (NCDC) about lead poisoning. The NCDC subsequently began working with tiro vendors to look into safer alternatives to using galena, and also initiated public health outreach efforts to raise awareness about lead in tiro and other sources. “The problem is not unique to Nigeria,” Woolf says. “Many countries in Africa and the Middle East lack formal systems for regulating or labeling cosmetics. When we saw the eyeliner, there was no label, no warnings, and no way to trace where it came from.”

In one sentinel case, a container of unlabeled diaper powder sent from Malaysia was found to consist largely of lead oxide. Credit: ©iStockphoto.com/ThitareeSarmkasat.
In 2017, Morman, Plumlee and Woolf collaborated on a sentinel case of a 9-month-old girl with blood lead levels as high as 18 µg/dl. “After [the medical investigation team] checked the family’s house and yard and found no lead, they asked the girl’s parents to bring in all the products they were using on the child for testing,” Plumlee says. “Sure enough, they were applying some kind of unlabeled, unbranded diaper powder the child’s grandparents had sent over from Malaysia. When we analyzed [the powder] we found it was very high in lead, and mineralogical studies showed that it was primarily lead oxide.”
The primary pathway for lead intoxication is via ingestion into the digestive system, where the metal is absorbed into red blood cells and circulated throughout the body. Woolf and his colleagues were curious whether urine could solubilize the lead in the diaper powder such that it might be more readily absorbed through the skin.
“In vitro bioaccessibility studies are a relatively easy and inexpensive way to identify how lead is being absorbed in the body,” Morman says. In such studies, researchers take samples of simulated gastric fluid or lung fluid and test the solubility of the particular type of lead involved. In the case of the Malaysian diaper powder, Morman developed a novel simulated urine bioaccessibility test, which revealed that the lead oxide was highly bioaccessible in simulated gastric fluids, but only slightly so in simulated lung fluids and even less so in simulated urine. The results suggested that the primary lead exposure routes for the 9-month-old were ingestion via hand-to-mouth transmission and ingestion of inhaled dusts cleared from the respiratory tract, rather than absorption of dissolved lead through the skin.
Four weeks after discontinuing use of the powder, the infant’s blood lead level had dropped to 8 µg/dl. The team followed up with Malaysian authorities, but the lack of identifying information on the unlabeled powder stymied efforts to track it to the source. “Cases like this make you worry about how many people are being exposed to lead through these unlabeled products in the country of origin,” Plumlee says. “It’s a pathway that’s very difficult to follow, and even harder to stop.”
In some instances, enough information can be gleaned about a lead-containing product to stop its distribution at the source. In a case at Boston Children’s Hospital, a 12-month-old child of Thai descent presented with a blood lead level of 61 µg/dl, which warranted immediate chelation therapy. The alarmingly high levels were tracked to the daily application of a black tongue powder called Ya Kward Pak (meaning “medicine to be applied to the mouth” in Thai) for the first seven months of the child’s life.
“This ethnic remedy was applied to the tongue to absorb toxins … and preserve the infant’s health,” Woolf’s team wrote in a 2008 study in Clinical Toxicology. Spectrometric analyses of the tongue powder showed lead in excess of 109,000 ppm, in the form of lead oxide.
The team contacted two poison control centers and government agencies in Thailand to launch a public health inquiry and recall. The Ramathibodi Poison Center, the Siriraj Poison Control Center, and the Thai Food and Drug Administration tracked the powder to a Chinese traditional medicine shop in Bangkok. Testing of several kinds of tongue powders and other remedies for sale in the shop revealed that many of the products had lead levels in excess of 9,500 ppm. The shop was ordered to stop selling the contaminated powders and was later closed for additional violations. The Thai FDA also conducted a press conference alerting the public in Bangkok to the dangers of lead and other heavy metals in traditional products, and the poison control centers opened a 24-hour hotline to handle cases of suspected infant poisoning.

A circa 1970 poster from the U.S. Department of Health, Education and Welfare (now the Department of Health and Human Services) warns of the hazard to children from lead paint. Nowadays, while many parents are generally aware of the dangers posed by lead, the metal still lurks in unexpected places. Credit: public domain.
The public health campaign that began in the 1970s to combat lead poisoning has dramatically reduced the incidence of childhood lead poisoning and cultivated widespread knowledge about the dangers of lead in the U.S. “These days, most Americans are familiar with the pitfalls of lead,” Landrigan says. This lead literacy is also due in part to the work of pediatricians and family doctors who “make a point to talk to parents about the common and uncommon sources, and the warning signs, of lead poisoning,” he says.
But while many parents are generally aware of the dangers posed by lead, the metal still lurks in unexpected places. And people who have immigrated to the U.S. in recent years may not have had the same experience with public health campaigns about the dangers of lead. A limited amount of research has shown a greater incidence of elevated blood lead levels in immigrant children. And, at least anecdotally, lead literacy seems less widespread among immigrant populations, although few studies have sought to quantify this knowledge gap.
“I do think there’s a gap in understanding in many international cultures that haven’t had the benefit of decades of public health campaigns,” Woolf says. “As physicians, we have to stay vigilant and remember that not everybody is well versed in the dangers of lead or the threat that unlabeled or mislabeled products might [pose] to children.”
Atypical lead poisonings sometimes spark international ripple effects, with dangerous products being tracked back to their place of origin and distribution stopped at the source. “Earth scientists can play an important role in these cases by helping doctors figure out what forms of lead somebody has been exposed to and pinpoint the pathway of intoxication,” Plumlee says. “Collaboration is key to solving these medical mysteries and figuring out how to keep them from happening again.”
© 2008-2021. All rights reserved. Any copying, redistribution or retransmission of any of the contents of this service without the expressed written permission of the American Geosciences Institute is expressly prohibited. Click here for all copyright requests.