
by Tiffany R. Jansen Thursday, June 16, 2016

Researchers at the Rich Earth Institute are experimenting with using urine-derived fertilizer to grow crops. Here, institute researcher Abe Noe-Hays adds sanitized urine fertilizer to lettuce. Credit: Marcin Szczepanski.
Whether you’re enjoying a juicy burger from a fast-food drive-thru or digging into a brightly colored salad at a backyard barbecue, your meal likely started life on a farm. Each day, agriculture around the world produces an average of 27.3 million tons of food, enough to feed Earth’s current population of about 7.3 billion — although not all of that food reaches people.
By 2050, researchers predict Earth will host more than 2 billion additional people, and to meet future demands, food production will need to increase significantly. To accomplish that, we’re going to need a whole lot more fertilizer.
Therein lies a problem. One of the primary nutrients in commercial fertilizer, along with nitrogen and potassium, is phosphorus. Phosphorus has no substitute as a nutrient and cannot be synthesized, but must instead be mined from existing sources, which, by some accounts, may have already reached peak production.
So what happens if current global resources of phosphorus run low or become inaccessible? Where might we find the necessary phosphorus so that food production can keep pace with humanity? According to some researchers, some of it could come from a surprising source: human urine.
Phosphorus is fundamental to life: It is part of the structural framework of DNA and RNA, it’s present in all cells, and it is the main component of bone and tooth enamel. It’s also essential to plant growth; the more phosphorus, the faster the growth and the higher the crop yield.
Phosphorus has been recognized as an important nutrient for millennia. From as long as 40,000 years ago until the late 1700s, Australian hunter-gatherers are known to have burned controlled patches of land, a process that converted phosphorus in the soil into bioavailable forms that could be absorbed by crops more easily. The Incas in Peru used bird guano — a fossilized and hardened material that is a combination of the bones of birds and fish, mixed with bird stool — as a phosphorus fertilizer for 2,000 years, and bat guano was used to treat fields in Japan during the Edo Era, from 1603 to 1868. Animal waste was widely used as phosphorus-rich fertilizer throughout Europe and Asia, dating back to the Middle Ages. Americans began harvesting phosphorus first in “bone mills” in 1830, grinding up the bones of animals like bison and spreading the remains on fields. In 1840, it was confirmed that phosphate, a phosphorus-containing salt, was affecting plant growth, and the hunt for phosphate sources was on.
Soon thereafter, Europeans and Americans started using phosphate from the Peruvian guano. But large concentrations were only found in Peru and on islands in the South Pacific, and these deposits were depleted by the late 1800s.
Meanwhile, phosphate rock deposits were found in Charleston, S.C., as well as along the Eastern Seaboard from New Jersey to Florida. Soon after the Civil War, northern investors convinced plantation owners in Charleston, who needed money for reconstruction, to begin selling off phosphate rocks, creating a new industry in the ravaged Southern city. The industry only lasted a couple of decades, however, and in 1889, a prospector named Albert Richter working out of Salt Lake City discovered the massive Phosphoria Formation, which lies mostly in Idaho but extends into Montana, Nevada, Utah and Wyoming. Deposited in a shallow Permian sea, the organic-rich black shales of the Phosphoria and related formations form the Western Phosphate Field, the most abundant phosphate deposit in the United States.

Phosphate mine near Flaming Gorge, Utah. The U.S.'s largest phosphate reserves are in the Western Phosphate Field in the northern Rocky Mountains. Credit: Jason Parker-Burlingham, CC BY 2.0.
Today, ores extracted from open pit mines in the Phosphoria produce both phosphate and phosphorus, with just two states — Idaho and Utah — accounting for about 20 percent of the U.S.’s annual phosphate output. Phosphate rock is also mined in Florida and North Carolina, which together produce the remaining 80 percent of the U.S.’s total domestic output, according to the U.S. Geological Survey (USGS) 2016 mineral commodity summary on phosphate.
The dramatic expansion of global agricultural production that has occurred over the last 75 years — a period called the Green Revolution, which coincided with a 50 percent increase in worldwide population — was made possible by the production of commercial fertilizers. These in turn were made possible by mined phosphate and synthesized fixed nitrogen, the latter of which came about after the 1909 discovery of a method to manufacture ammonia (for which Fritz Haber and Carl Bosch each won a Nobel Prize). Today, food production accounts for 90 percent of global phosphorus use.
Phosphate rock reserves are finite. With reserves dwindling, miners are forced to extract phosphate from lower-quality ores, which means more mining, more purification and more energy inputs, all of which translate to greater costs.
Like peak oil, “peak phosphorus” is the point at which phosphate production will reach maximum output before beginning to decline. There is disagreement about when peak phosphorus will occur: Predictions range from as soon as 30 to 40 years from now to 10 times that long. But a phosphate shortage isn’t the only issue.
“It is as much about geopolitical scarcity as it is physical scarcity,” says Dana Cordell, co-founder of the Global Phosphorus Research Initiative and research principal at the Institute for Sustainable Futures at the University of Technology in Sydney, Australia.
Economically minable deposits of phosphate rock are found in just a handful of countries. Together, Morocco, China, Algeria, Syria and South Africa control 88 percent of the world’s phosphate reserves, according to USGS. Morocco alone controls about 75 percent of the world’s high-grade reserves and is the second-largest producer, behind China. The United States, which has the ninth-largest reserves in the world but is the third-largest producer behind China and Morocco and one of the biggest consumers of phosphorus, is expected to run out in as few as 20 years. More and more of the country’s phosphate is now being imported, especially from Morocco.
This quasi-monopoly on phosphate rock gives producing countries power over market prices. China, for example, imposed a 135 percent export tariff on phosphate in the wake of the 2008 economic crisis, when global phosphate prices shot up by 700 percent from $50 to more than $400 per ton.
Though phosphate prices have since fallen — hovering between about $100 and $200 per ton since mid-2009 — net importers remain vulnerable to economic uncertainties and market forces influenced by producers. And many farmers around the world are unable to afford fertilizer, with up to a billion without access to fertilizer markets.
thout new sources of phosphate to rely on, some researchers are seeking ways to stem leakage in the human phosphorus cycle — the path that phosphorus follows through the ecosystem once it is liberated by mining. Steps in the cycle include application to plants during farming, run-off into waterways, and consumption and excretion by humans, among others.
Only about 20 percent of mined phosphate actually makes it into the food we eat, according to a 2011 BioScience study by Daniel L. Childers of Arizona State University. Much of the rest is lost through inefficiencies in the mining, purification and transportation processes, as well as in the production, distribution, application and runoff of phosphate fertilizer.
One option to reduce loss is to reduce demand for phosphorus. This can be done by switching to less phosphorus-intensive diets — meaning fewer meat and dairy products, and more local and in-season foods — and minimizing food waste. According to the U.S. Environmental Protection Agency, Americans threw out an estimated 35 million tons of food in 2012. Demand can also be curtailed in agriculture, by planting cover crops or using nutrient management programs to ensure more efficient use of fertilizer and to minimize runoff.
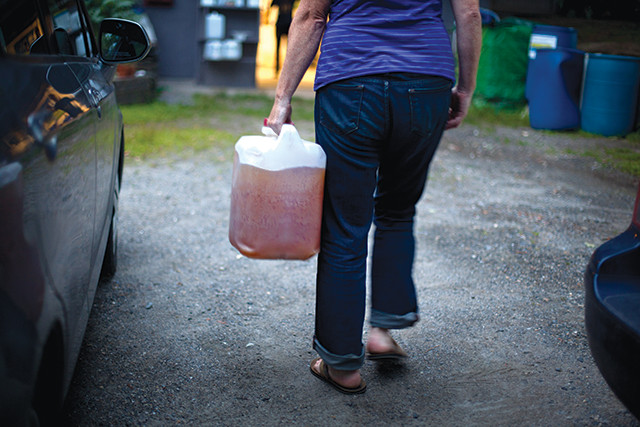
A urine donor brings her donation to a depot at the Rich Earth Institute. Credit: Marcin Szczepanski.
Another option is to recycle it. “The great thing about phosphorus [is that] it’s not like energy or oil [where] once you burn it, it’s gone. You can use it over and over again,” says James Elser, an ecologist at Arizona State University and co-founder, with Childers, of the university’s Sustainable Phosphorus Initiative, which seeks ways to close the human phosphorus cycle.
Animal, crop and food wastes are the primary sources from which phosphorus can be recycled. But there’s another ubiquitous and readily available source that researchers are considering: human waste.
Humans expel an estimated 3 million metric tons of phosphorus annually. That’s enough to meet almost a quarter of the global demand. Fifty to 60 percent of the phosphorus in human waste is in urine, which is typically very low in pathogens. A year’s worth of urine from just one person contains enough phosphorus to produce at least half of their annual food requirement.
With the possibility of a phosphate crisis looming, attention has turned to the power of pee as environmentalists and scientists look to urine as the future of fertilizer.
“Urine is a perfect fertilizer,” says Tove Larsen, a senior scientist studying urban water management at Eawag in Switzerland.

Hayfields treated with urine have been shown to produce significantly higher yields than unfertilized control plots, and equivalent yields to plots treated with synthetic fertilizer. Credit: Abe Noe-Hays.
Indeed, plants have been shown to grow extremely well on human urine. In an experiment by the Vermont-based Rich Earth Institute, which promotes recycling human waste to meet resource needs, hayfields treated with urine had significantly higher yields than unfertilized control plots, and equivalent yields to plots treated with synthetic fertilizer. Finnish researchers, meanwhile, found that red beets fertilized with human urine grew 10 percent larger on average than those treated with mineral fertilizer.
“Peecycling,” as it’s sometimes called, has also been tested in public initiatives over the last few years. The organizers of Denmark’s famed Roskilde music festival, for example, plan to make beer from malted barley fertilized by patrons’ urine. In 2015, SAIL Amsterdam, the largest free public event in the world, gathered urine in special urinals to be processed into fertilizer for “green roof” projects. And Treavor Boyer, an environmental engineering professor at the University of Florida, has his sights set on treating the school’s football field with urine from sports fans, captured in specially designed toilets installed at the stadium.
Although the idea of human waste as fertilizer is nothing new — it was widely practiced in ancient Greece, much of eastern Asia and even in Tudor England — currently, only 10 percent of human excrement globally is intentionally returned to farm fields, either as wastewater or treated sewage sludge.

Seth True from Best Septic Service of Westminster, Vt., pumps human urine for transport to the Rich Earth Institute's participating farms. Credit: Abe Noe-Hays.
Some of the reluctance to recycle human waste can be attributed to the “yuck factor,” says Krista Wigginton, an assistant professor of civil and environmental engineering at the University of Michigan. Terms like “recycled sewage” and “toilet-to-tap” conjure, what are to some, disturbing thoughts and unsavory images.
Most people think of excretion in terms of waste rather than as a resource, Wigginton says, and “that there is nothing valuable in their feces and urine.” The tendency is to flush and forget, never realizing the value in what’s going down the drain. Most are also unaware of the rigorous treatment and pasteurization processes that can render human waste perfectly safe for use as fertilizer. Last year’s blockbuster movie “The Martian” may help change that view — the protagonist grew potatoes on Mars using his waste as fertilizer — but there’s still a long way to go both in terms of the infrastructure and public perception.

At Eawag, urine goes through a sanitization process in which other nutrients such as magnesium chloride can be added to extract solid phosphorus, and the sanitized urine is thoroughly tested for safety before it is applied to gardens or agricultural lands. Credit: top two: Yvonne Lehnhard, Eawag; bottom two: Martin Koller, Eawag.
Each time you flush the toilet, along with solid and liquid waste, you send 6 to 13 liters of water down the drain. Upon arrival at a treatment plant, bacteria break down contaminants, forming sewage sludge, which is separated from the treated wastewater. The wastewater is then sanitized with chlorine to kill remaining microbes.
Traditional sewer systems and wastewater treatment plants do a fine job of cleaning wastewater — toilet water in the U.S. is routinely cleaned to drinking water standards. But even though some systems and plants are capable of capturing and recycling phosphorus, most are not efficient at doing so.
Wastewater treatment plants in some communities are equipped with nutrient removal technologies to prevent nutrient pollution of waterways. These systems pull phosphorus from the wastewater, either biologically or chemically. The extracted nutrients are then sometimes returned to agricultural soil as part of sewage sludge. Other facilities only clean the waste stream as much as necessary to meet sanitary regulations before releasing it into surface waters, where much of the phosphorus is lost.
While nutrient removal technology can be added to all treatment plants, it is expensive, and separating the nitrogen and phosphorus, most of which is contained in urine, from diluted wastewater at plants is costly and time-consuming. “It is very energy intensive and very chemically intensive,” Wigginton says.
A better way to maximize phosphorus recovery from human waste, according to Larsen, is to separate urine and feces at the source. Since the mid-1990s, engineers have been working to design toilets that would separate the waste streams as they are excreted. Referred to as “separation,” “urine diversion” or “no-mix” toilets, they can be either wet or dry and look, on the outside, like a typical toilet. The inside of the bowl, however, is divided into two collection areas; one at the front for urine and another at the back for feces.
Less water is required for the diversion system, Wigginton says, and results in a more concentrated stream of urine. This makes treatment of the waste stream and reclamation of phosphorus and nitrogen more efficient.
t the University of Michigan, urine-diverting flush toilets are being installed in the women’s restrooms of the campus’ civil and environmental engineering building to test the diversion method in a public setting. Plumbing there is set up in such a way that the feces are transported to the treatment plant via the traditional sewer system. Urine follows a separate pipe leading to a collection chamber in the basement.
The collected urine is then stored for six months, during which the pH and ammonia concentration rise, deactivating pathogens and killing off unwanted organisms. At this point, the urine is safe to use as fertilizer and can be applied directly to fields.

Application of urine fertilizer at the Rich Earth Institute's Whetstone Valley Farm (above) in Brattleboro, Vt. Credit: Abe Noe-Hays.

The U.S. Environmental Protection Agency funded experiments at the Rich Earth Institute using urine to grow lettuce. Credit: Mike Earley.
Solving engineering issues in the bathroom may turn out to be a relatively easy part of the problem. Implementing systems designed to sort solid and liquid wastes on large scales will be more challenging.
Were the will and the funding available, these systems could be particularly useful and easier to implement in the developing world, where wastewater treatment has not yet been implemented or technologies are unadvanced, according to a 2011 review by James R. Mihelcic of the University of South Florida, published in Chemosphere. In the developed world, however, changing over to such a system would be a monumental undertaking.
If we decide to go that route, Wigginton says, infrastructure will be the biggest hindrance. “Right now, our sewage systems, our pipes and buildings, our fittings, everything is designed for combining our wastewater,” she says. Digging up sewers to create separate pipelines is going to be costly.
As a start, Larsen recommends installing waterless urinals and urine-diverting flush toilets in new buildings and replacing traditional toilets during major renovations of older structures. Dormitories and apartment buildings are also possible starting points, Wigginton says.
Larsen agrees that changing everything over at once is simply unaffordable. “If you do something like this,” she says, “you do it slowly.”
Back at the University of Florida, Boyer and his team are hard at work developing a source separation technique to harvest the essential nutrients phosphorus, nitrogen and potassium from the urine of the approximately 90,000 Gator fans that fill Ben Hill Griffin Stadium each game day. “Right now, urine collection at stadiums is [still just] an idea,” Boyer says, “but we think it’s feasible from our preliminary research.”
© 2008-2021. All rights reserved. Any copying, redistribution or retransmission of any of the contents of this service without the expressed written permission of the American Geosciences Institute is expressly prohibited. Click here for all copyright requests.