
by Mary Caperton Morton Tuesday, September 13, 2016
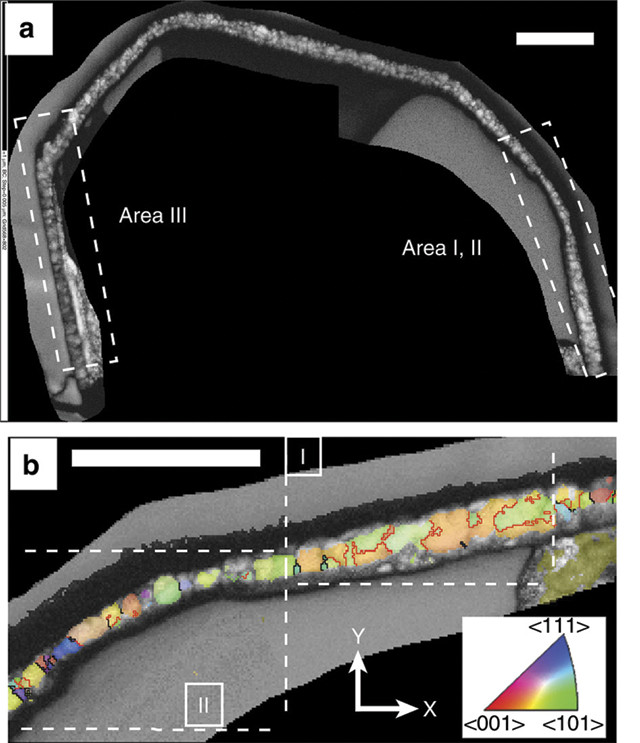
Researchers found a thin magnetite corona — the dappled band shown in (a) and with individual grains colored in (b) — around a sulfide inclusion in a diamond from Botswana, evidence of a diamond-forming process called redox freezing. Scale bars are 0.5 micrometers. Credit: Jacob et al., Nature Communications, June 2016, CC BY 4.0.
Not all diamonds are gems. Those found bearing imperfections or inclusions, known as bort or junk diamonds, are often demoted to industrial uses such as in diamond-tipped drill bits. They also sometimes find their way to scientists interested in using the inclusions to study how such stones form. Now, a team studying a junk diamond from Botswana with a large black sulfide inclusion has uncovered evidence of a previously unrecognized diamond formation process that occurs in the deep mantle.
Diamonds form in Earth’s mantle as a result of high-temperature, high-pressure reactions between carbon-bearing fluids and minerals like olivine. Studying diamonds can shed light on the workings and composition of the deep mantle, says Dorrit Jacob, a mineralogist and geochemist at Macquarie University in Australia and lead author of a new study in Nature Communications.
“Botswana is an interesting place to study diamonds because the volcanoes [there] bring up all sorts of rocks and diamonds from Earth’s mantle,” Jacob says. The diamond she and her colleagues used in the study was a “lucky find,” she says, as it helped illuminate a diamond-forming process called redox freezing that had only been hypothesized.
To study the microstructure of the diamond and its black, iron-nickel sulfide inclusion, Jacob’s team turned to a pair of methods borrowed from materials science. The first, called focused ion beam milling, is used to make ultra-thin slices of solid materials. The technique produced slices so thin that electrons could pass through the diamond, revealing the nano-structure of the sulfide inclusion trapped inside. They next used the Transmission Kikuchi Diffraction (TKD) method, named for the Japanese physicist Seishi Kikuchi who first observed and explained electron diffraction in a thin film of mica, which involves identifying miniscule mineral grains based on how they uniquely diffract electrons. Using TKD, the researchers found a sort of halo, or corona, of the mineral magnetite just a few ten-thousandths of a millimeter thick surrounding the sulfide minerals inside the diamond. This pattern points to redox freezing, in which oxidation of the sulfide minerals into magnetite triggers the precipitation of diamond around the inclusion.
“People have looked at these inclusions before but they didn’t have the technique to look at the microstructure of the corona of magnetite around the sulfide,” says Steven Shirey, a geochemist at the Carnegie Institution for Science in Washington, D.C., who was not involved in the new study. “That’s the key to this new finding.”
Jacob says it is not yet clear how common this redox freezing reaction might be in the deep mantle, although sulfide inclusions are some of the most common inclusions found in diamonds, even though sulfide itself is relatively rare in the deep mantle. “It’s always been a bit of a mystery why we see so many sulfide inclusions in diamonds, but with this redox method it seems that sulfide is present because it’s a key player in the formation of the diamond itself,” she says. The next step in the research will be to find and test more sulfide-bearing diamonds using the TKD method, Jacob says.
Although it’s technically conceivable that the redox freezing reaction could be used to grow diamonds in the lab, it’s unlikely that will happen, Shirey says, because it’s much less efficient than current methods used to manufacture synthetic diamonds. “Growing diamonds was a breakthrough in the 1950s, but these days it’s not too difficult.”
© 2008-2021. All rights reserved. Any copying, redistribution or retransmission of any of the contents of this service without the expressed written permission of the American Geosciences Institute is expressly prohibited. Click here for all copyright requests.