
by Mary Caperton Morton Thursday, October 12, 2017
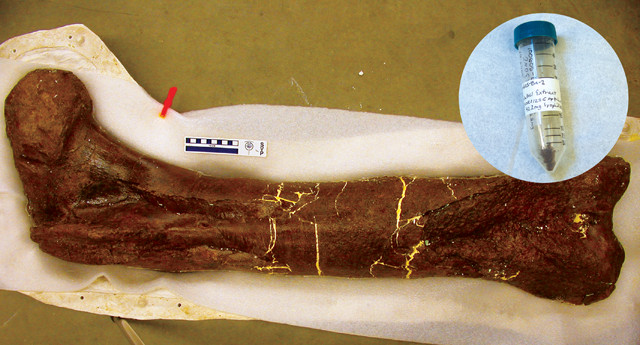
Mary Schweitzer, of North Carolina State University, and her colleagues first reported the discovery of soft tissue in a Tyrannosaurus rex femur in 2005. The vial contains a sample of the bone. Credit: both: ©Science.
In the early 1990s, “Jurassic Park” hatched a wild idea: that dinosaurs could be reincarnated using tiny amounts of preserved DNA. In the bestselling books and blockbuster movies, the premise hinged on extracting fragments of dinosaur DNA from ancient blood-sucking insects preserved in amber, filling in the genetic gaps with snippets of modern amphibian DNA and hatching the engineered dinosaurs out of ostrich eggs. In reality, however, while many amazing things have been found in fossilized tree sap, DNA isn’t one of them.
“Jurassic Park” fans saw a glimmer of hope in 2005, when a team of molecular paleontologists led by Mary Schweitzer of North Carolina State University reported finding soft tissue preserved inside a 68-million-year-old Tyrannosaurus rex femur. DNA is notoriously delicate, and dinosaur DNA itself has not been found — a possibility Schweitzer calls extremely unlikely since the oldest DNA recovered to date is less than a million years old and nonavian dinosaurs died out 66 million years ago. However, in the decade since that initial find, Schweitzer’s team has unearthed mounting evidence that soft tissues, such as blood vessels, collagen and other proteins, whose long, folded chains of amino acids make them much more robust than DNA, can survive more than 66 million years of degradation.
If 68-million-year-old dinosaur soft tissue can really be recovered — a claim that has been met with much skepticism — it may help paleontologists answer some long-standing questions about dinosaur physiology, such as whether the behemoths were cold- or warm-blooded. The existence of ancient preserved soft tissues would also change how we think about fossilization and the handling and storing of fossils: After all, if soft tissue can be recovered from ancient fossils, we need to be more careful in how we extract and preserve them. Living, roaring tyrannosaurs may not be the future, but perhaps molecular paleontology is.

Schweitzer working in a lab at Montana State University. Her team has found evidence for soft tissues in about one-third of the several dozen dinosaur specimens they have studied. Credit: Kelly Gorham, Montana State University.
The young field of molecular paleontology was thrust into the international spotlight in 2005 when Schweitzer’s team announced in Science their discovery of T. rex soft tissue. The tissues were found by accident: After some broken femur fragments were dissolved in a weak acid solution, a scrap of oddly stretchy material was left over. Under a microscope, the scrap showed what appeared to be bifurcating blood vessels, cellular structures similar to those found in bone cells of modern animals, as well as fibrous bone matrix tissue and small, round red structures resembling red blood cells. The 68-million-year-old tissue had all the hallmarks of collagen — the main component of connective tissue and one of the most abundant proteins in living animals. “When I first saw [the tissues], my first thought was, ‘Those aren’t supposed to be there.’ Back then, I didn’t think soft-tissue preservation was possible either,” Schweitzer says.

Credit: K. Cantner, AGI.
As far as biomolecules go, collagen is tough, but nobody thought it could be that tough. The oldest nondinosaur collagen recovered to date came from a 3.5-million-year-old camel fossil found in the high Arctic of Canada. The unique geochemical conditions required for long-term preservation of proteins like collagen are still unknown. In fact, the process of fossilization in general remains somewhat mysterious, in part because the rigors of geologic time are impossible to reproduce in the lab. In conventional fossils, just hard parts such as bones, teeth and shells remain; soft parts are typically lost to predation or decay before being fossilized. In exceptional cases, such as the fossils of the Burgess Shale in British Columbia, delicate soft-tissue structures, such as gills or stomach contents, are mineralized and preserved as rock, but what Schweitzer found was even stranger: actual soft tissue that had somehow escaped scavengers, rotting and the ravages of time for 68 million years.
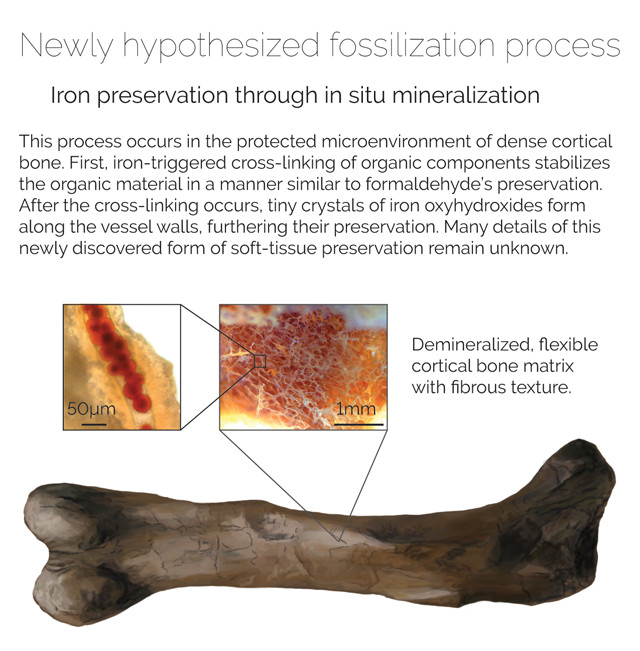
“Living bone is made up of minerals plus organic matter,” Schweitzer says. After death, as the organics, such as collagen, degrade, they leave behind voids in the bone at the molecular level. During fossilization, those voids fill with minerals, which help preserve the bone for millions of years as a fossil. “The leaving of the organics is part of what allows the fossil to form,” she says. “But in reality, this textbook process of fossilization doesn’t explain all the various modes of fossilization we see, some of which we have no idea about how they happen.”
Since their initial discovery in 2005, Schweitzer’s team has published dozens of studies identifying collagen, blood vessels, hemoglobin, the proteins actin and myosin, and molecules called histones that help package DNA, in the 68-million-year-old T. rex and in an 80-million-year-old hadrosaur, both found in Montana.
When identifying unusual tissues, Schweitzer starts with high-resolution light microscopy to examine samples for evidence of deep age: If tissues are actually many millions of years old, certain levels of damage are expected. “Does it look altered from a living tissue? Is it possible these are original molecules?” Schweitzer asks. Then, she turns to antibodies, which recognize and bind to certain types of proteins present in a sample. “Antibodies are extremely specific. If I can get a signal, then it justifies the added expense and destruction of [the sample required for] mass spectrometry sequencing,” she says. Mass spectrometry is the definitive method used to identify and characterize both modern and ancient proteins.
The team’s most recent study, published this year in the Journal of Proteome Research, revisited collagen they recovered from the hadrosaur. In a 2009 study, Schweitzer’s team identified three fragments, or peptides, from Type 1 collagen. Peptides are made up of amino acids — the building blocks of protein — and each of the fragments was about 15 amino acids long. As read by a mass spectrometer, the three fragments appeared to be similar to collagen 1 peptides found in living reptiles. For the 2017 study, the team worked with new samples from the same hadrosaur and used updated extraction methods and more sensitive mass spectrometry to identify eight peptides: six new and two that matched the 2009 fragments. The new fragments were found to line up most closely with collagen found in modern birds — the surviving dinosaurs.
“This latest study is an effort to show that our data are repeatable,” Schweitzer says. “Everything was different, except for the source dinosaur. We worked in a different lab, with different instruments, using different extraction buffers and different analytical tools, and we still got the same sequences.”
Ideally, other groups would also validate the findings, but not many places are equipped to study such ancient proteins, says Elena Schroeter, a postdoctoral researcher at North Carolina State University and lead author of the 2017 study. “An important step in the scientific process is replication of data in other labs. That’s been one of our major problems: This is high-risk research, it’s hard to get funding, and it is very expensive,” she says. “You need to be able to dedicate a lab exclusively to ancient proteins to keep everything isolated, to avoid the possibility of cross-contamination, and not many places are set up for that.”
The new study is “the most convincing evidence I’ve seen yet that these proteins really are from dinosaurs,” says Enrico Cappellini, a professor of paleoproteomics at the Natural History Museum of Denmark in Copenhagen. “[Schweitzer’s] lab is very careful about contamination,” he says. “They’re doing everything right in keeping the ancient stuff totally separate from modern stuff. My inclination is that their data are authentic, but there’s still room for improvement. I can understand why many of my colleagues are not yet convinced.”

Bone cell (left) and red blood cells (right). The brown cells (in focus) are surrounded by a white fibrous matrix from the B. canadensis specimen that Schweitzer's team studied. Credit: both: courtesy of Mary Schweitzer.
Cappellini says he would like to see the extraction procedure simplified. Schweitzer’s technique “has a lot of steps, and with each step, you get a loss of peptides. With a less convoluted procedure, they might be able to recover a higher number [of peptides] and other labs might be able to follow the technique,” he says.
One of the most vocal critics has been Matthew Collins, a biochemist at the University of York in England who specializes in recovering ancient proteins. “Mary Schweitzer is either doing something very right or she’s doing something very wrong,” Collins says. At this point, the most convincing data would come out of an independent lab, he says. “I would be really happy if other labs could replicate [Schweitzer’s] data. [My lab has] tried and we’ve been unsuccessful. So far, they’re the only ones who have been able to do this.”
Much of the criticism from Collins and others involves questions about contamination, either from bacterial biofilms that invaded the fossil long ago and produced the soft-tissue-like materials, or from modern contaminants introduced in the lab. In May, Michael Buckley of the University of Manchester in England and his colleagues published a study in Proceedings of the Royal Society B that highlights the difficulties of discriminating dinosaur proteins from modern cross-contaminants. Buckley and his team extracted collagen from modern ostrich bone and identified protein sequences identical to those found by Schweitzer’s team in the hadrosaur in their January study and the tyrannosaur in the original 2005 study. The findings were “a bit suspicious,” Cappellini says. They mean “that either 80-million-year-old dinosaur proteins are identical to those found in modern ostriches or that [Schweitzer’s] lab was contaminated with ostrich proteins.”
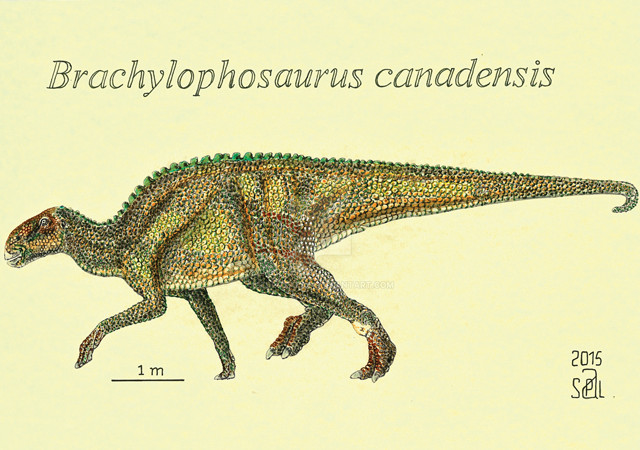
Schweitzer's team recovered soft tissue from a B. canadensis fossil found in Montana. Credit: Pedro Salas.
Schweitzer says it’s understandable that some of the hadrosaur proteins will be identical to those found in modern ostriches — because collagen is known to be a highly conserved molecule across a diverse array of species. Dinosaurs and their modern relatives — birds — could share some of the same protein sequences, she says. “Many molecules are conserved across species, over millions of years. They work, so they remain unchanged by evolution.” In fact, she says, some amount of sequence overlap would be expected, so the similarities between the hadrosaur and ostrich peptides don’t mean there was contamination in her team’s samples.
The limited availability of established protein sequences is one of the big challenges in this field, she says. “We are dealing with the limitations of an emerging discipline. When you have a 66-million-year-old protein, what do you compare it to?” Currently, Schweitzer’s team has to settle for comparing protein sequences to those of extant bracketing species such as birds and crocodiles. “But the databases are very limited for creatures that are not mammals. These animals [ostriches and crocodiles] were not represented in existing databases until we put them there. "

Field emission scanning electron microscope images of demineralized ostrich bone matrix (left) versus B. canadensis bone matrix (right). Both show osteocytes (bone cells). Some of the ostrich proteins are identical to the B. canadensis proteins. Credit: both: courtesy of Mary Schweitzer.
Schweitzer and her colleagues feel they have demonstrated that contamination isn’t undermining their sensational findings, but they have not yet figured out how ancient tissues can persist for millions of years. “That’s the million-dollar question. How on Earth is this possible?” says Tim Cleland, a molecular biologist at the Smithsonian’s Museum Conservation Institute in Washington, D.C., who has co-authored papers with Schweitzer, including the 2017 study.
“I have yet to hear a plausible explanation for how soft tissues can be preserved for this long,” Collins says, citing the huge leap of time from 3.5 million years to more than 66 million years. “The more I understand about it, the more difficult it is to believe it can happen,” he says. “There may be something very special about these fossils, but for me they’re defying basic chemistry and physics. I’d like to see better comprehension of what happens when proteins decay and how decay might be suspended indefinitely.”
In 2013, Schweitzer’s team offered a hypothesis: The preservative could be iron. Tissues from both the tyrannosaur and the hadrosaur they studied were found to be chock-full of iron nanoparticles. Iron is abundant in living organisms, especially in the blood, where it makes up part of the oxygen-transport protein hemoglobin. Iron is notoriously bioreactive, so much so that organisms must take biochemical precautions to prevent excess iron from causing tissue damage. But while iron can be detrimental to living tissues, it can act like a preservative in dead tissues, Schweitzer says.
“When a dinosaur dies, it has a boatload of iron in its blood and muscles. And when that iron is released into the carcass, it goes about cross-linking like crazy, tying proteins and cell membranes in knots, basically acting a lot like formaldehyde,” she says. Tissues treated with formaldehyde can sit in a jar on a shelf for decades, but decay is only delayed, not arrested. To demonstrate iron’s preservative capabilities, Schweitzer extracted blood vessels from modern ostrich bones and treated them with iron-rich hemoglobin. “The control [samples turned to] mush in three days, but the vessels exposed to hemoglobin are still sitting on a shelf in my lab at room temperature six years later,” she says. Collins says he remains skeptical. “Iron may slow down the decay process but it’s not clear how it could be arrested altogether,” he says.
The internal conditions of a carcass play a role in the initial stages of tissue preservation, but the external environment is crucial for fossilization. "Every fossil has its own unique geochemical environment," Schroeter says. And while paleontologists have a good idea where they should look for dinosaur fossils — in Mesozoic sedimentary rocks, usually in hot, dry places such as eastern Montana and northeastern Utah — they're still figuring out what conditions are more likely to support fossils with preserved soft tissues and how much might exist. "It's an open question, how much of this [soft-tissue] material is out there," Cleland says. Soft tissue in dinosaur fossils is rarely found, but that may be because "people rarely look" for it, Schroeter says. Credit: K. Cantner, AGI; inset figures modified from Schweitzer et al., Proceedings of the Royal Academy B, 2006.
It may be more common than we realize, Schweitzer adds. Her team has found evidence for soft tissue in about a third of the several dozen specimens they have studied. “We will probably find more if we look more,” she says.
Schweitzer’s team has been working on formulating a profile of burial environments that seem to be conducive to protein preservation. “Fossils that are found in sandstone seem to have better preservation than those that come from shale or claystone, but we don’t really know why that is yet,” Cleland says. “The only thing we know for sure is that the carcass needs to be buried very quickly after death.” The dinosaurs that Schweitzer’s team has worked with have been found partially articulated — a sign that the carcasses were not pulled apart by scavengers.
“You have to be pretty lucky to find a well-preserved dinosaur bone, let alone a whole dinosaur,” Schroeter says. “There’s a huge range of preservation out there. Some fossils have perfect histological structure and look just like pristine bone in terms of their microstructure, while other fossils look just like rocks.”
Once fossilized materials are removed from the ground, whatever equilibrium they were in can be disrupted. In an effort to preserve any remaining soft tissues and prevent contamination, Schweitzer’s team has developed a new approach to excavating and handling fossils. “It’s best to keep the fossils in the sediment they were in until you get them into a controlled lab setting,” Schweitzer says, even though this sometimes results in massively jacketed fossils that are a logistical nightmare to transport. “We can remove the fossil wearing gloves, without using any glues, using sterile instruments and containers in an isolated lab.”
Then, instead of being stored in museum drawers where they might be contaminated, the fossils are kept in sterile glass jars, in special cabinets isolated from other fossils or modern samples, in a room far removed from other specimens. The team is also developing better techniques for storing these samples long term. “Technology gets better and more sensitive by leaps and bounds all the time,” Cleland says. “We want these samples to be around in [a decade] and even longer.”
Testing for proteins requires destructive sampling, in which bits of the fossils are inevitably destroyed. “Plenty of curators aren’t big fans of destructive sampling,” Schroeter says. “I’d really like to test more specimens, but it can be hard to get people to let you grind up their fossils.”
Paleontology has long relied on scant clues gleaned from bones turned to stone, and the possibility of introducing molecular techniques into the field is tantalizing to some researchers. “Part of the power of molecular paleontology is the ability to look at dinosaurs from a different angle,” Schroeter says. “Morphology can get you pretty far, but there are a lot of things that bones can’t tell you.”
Even skeptics agree that molecular paleontology has great potential. “If soft-tissue preservation is possible, it would open vast areas of the fossil record to molecular analysis,” Collins says. “Honestly, I really hope [Schweitzer] is right.”

Many scientists are loathe to allow even tiny pieces of fossil, like this piece of rib from a Lufengosaurus, a sauropod, to be destroyed — a necessary process for molecular paleontology. Credit: Lee et al., Nature Communications, 2017.
A molecular approach could answer long-standing questions about the physiology of dinosaurs, such as how they regulated body temperature, how their cardiovascular systems were able to support such large creatures, and how they grew and reproduced, as well as resolve how various species were related to one another, how they spread around the planet, and how some lines evolved into birds. But the field has a long way to go.
One of the biggest hurdles is the difficulty of proving endogeneity — that the proteins recovered come from a dinosaur and not a bacterial or modern source (see sidebar, page 45). “When you are testing fossils, it is critical to use multiple methods to examine and reexamine, test and retest different aspects of the molecules we think we’re recovering,” Schweitzer says. “To get the field moving forward, we need to come to a consensus on which methods are the most efficient, and which methods can tell us the most with the least amount of [sample] destruction.”
Other teams have developed methods of testing for ancient preserved proteins that don’t require destructive sampling, but the methods’ reliability is uncertain and they provide limited information about samples. For example, Yao-Chang Lee of the National Synchrotron Radiation Research Center and National Central University in Taiwan and colleagues published a study in Nature Communications this year in which they claim to have recovered collagen from a 195-million-year-old long-necked Lufengosaurus. The researchers described using nondestructive infrared spectroscopy to identify the protein. But there’s a flaw with this method, Cappellini says. “With this technique, you can see a chemical signature of what might be a protein, but it’s not a clear fingerprint like you get with mass spectrometry.” And that’s just not good enough, he says. “On controversial samples this ancient, it’s necessary to have unquestionable evidence.”

Recep Avci (standing) and Jahson Suo look at images of soft tissue from dinosaurs under a microscope. Credit: Kelly Gorham, Montana State University.
After weathering years of controversy, Schroeter says she is beginning to feel like a mouse running endlessly on a wheel: “Some of us in Mary’s lab feel like we’re stuck continuously arguing that these proteins can persist, even though we’ve shown repeatedly that they do,” she says. “We want to get beyond these same arguments and focus on what we can learn from these tissues.”
Schweitzer says she chooses to respond to her critics by working harder. “The doubters push me to do everything the best I can do it,” she says. “I want to make this work last for the ages.” Neither Schweitzer nor Schroeter are holding their breath for a single piece of evidence that will satisfy their critics once and for all. “When making a scientific argument … you want a bunch of different lines of evidence that all point toward the same thing,” Schroeter says.
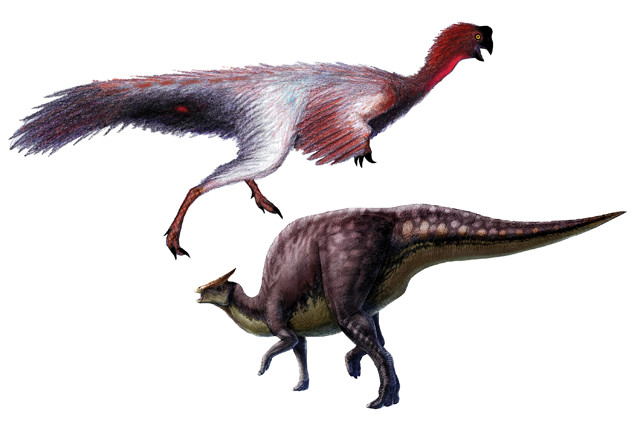
Whether it's finding blood vessels from hadrosaurs, like this Saurolophus (right), or determining the colors of feathers on dinosaurs like the Jiangxisaurus (left), researchers hope molecular paleontology can reveal many long-kept dinosaur secrets. Credit: right: L. Xing and Y. Liu, PLOS ONE, CC BY 2.5; above: Danny Cicchetti, CC BY 3.0.
Interest in the field is growing, but the number of labs set up for this kind of work remains limited, Schroeter says. “Paleontology has always been a little slow on the uptake in adopting new technology, but as more young people get interested, they’ll bring their tech savvy,” she says. “To be competitive, you have to constantly be thinking of new questions, new approaches, new ideas. This is how we’re going to raise the bar for paleontology.”
© 2008-2021. All rights reserved. Any copying, redistribution or retransmission of any of the contents of this service without the expressed written permission of the American Geosciences Institute is expressly prohibited. Click here for all copyright requests.