
by William H. Orem Thursday, January 5, 2012

Florida panther. National Park Service Photo by Rodney Cammauf
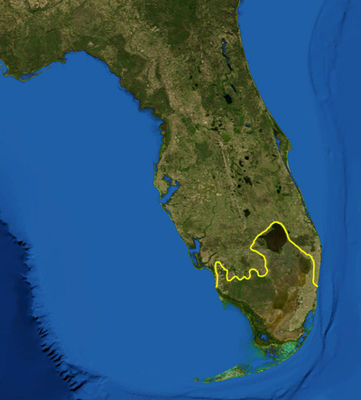
According to the World Wide Fund for Nature, the Everglades 'ecoregion' (south of the yellow line) includes Lake Okeechobee, the Everglades, Big Cypress Swamp, the Atlantic Coastal Ridge, the estuarine mangroves of the Ten Thousand Islands and Florida Bay. NASA

Aerial view of the Everglades. National Park Service

Freshwater prairie. National Park Service Photo

American Alligator. National Park Service Photo by Rodney Cammauf

Great Egret in Everglades National Park. National Park Service Photo by Rodney Cammauf
Pick Your Poison: If One Pollutant Doesn’t Take Down the Everglades, Another Might
As the sun rises over the vast Florida Everglades, the endangered Florida panther quietly stalks a white-tailed deer in the tall grass. A raccoon fishes for its breakfast of crayfish. A small flock of rose-colored waterfowl flies overhead, a reminder of the vast flocks of wading birds that once called the Everglades home. A couple of otters roll around in the water nearby, keeping a watchful eye out for ubiquitous alligators. Manatees swim silently below the surface in Florida Bay, at the southern end of the Everglades ecosystem. Suddenly, a tractor engine revs to life as a farmer prepares to harvest his sugarcane, and the noise of commuters driving to work on the Sawgrass Expressway disturbs the calm. Such is life in the Everglades, where modern civilization meets wild in a vast subtropical wetland.
Stretching nearly the width of southern Florida, the Everglades, or “river of grass,” extends more than 160 kilometers from Lake Okeechobee in the north to Florida Bay to the Gulf of Mexico in the south. The Everglades consists of a mosaic of habitats: ridges of thick sawgrass separated by open water channels, or sloughs, with a scattering of tree islands that rise slightly above the surrounding grass and water. The Everglades ecosystem is home to top predators such as black bears, panthers and alligators, as well as wading birds, many species of small mammals, including mice, rats and weasels, and other unique flora and fauna, some of which are endangered.
A combination of factors formed the Everglades about 5,000 years ago: The retreat of glaciers after the last ice age, subsequent sea-level rise, the flat landscape of southern Florida, and a change to a subtropical climate with increased rainfall led to the formation of a vast wetland. About 5,000 years ago, peat soil began to accumulate, formed from the remains of aquatic plants preserved in the waterlogged conditions. Rainfall and overflow from the Kissimmee River and Lake Okeechobee fed and maintained the wetland, with water flowing from the north and northeast to the southwest as a broad, shallow sheet. Because rainfall contributed most of the water, soils and water in the Everglades were low in nutrients, especially phosphorus, and other dissolved ions. The plants and animals that colonized the area became well adapted to survive in a low-nutrient, freshwater ecosystem.
By the late 1800s, modern civilization moved into the region and dramatically changed the face of the Everglades. Cities such as Miami and Fort Lauderdale and their associated infrastructure popped up in the east to accommodate the droves of people who wanted to enjoy southern Florida’s warm climate. Farmers created the Everglades Agricultural Area just south of Lake Okeechobee to grow sugarcane and vegetables. The federal government constructed canals and levees to preserve farms and homes from the annual floods the region had endured for millennia.
This development altered the Everglades’ hydrology, causing some parts to flood while other parts thirsted for water, especially during the dry season, which lasts from November to June. In the early 1960s, officials carved large impoundments, called Water Conservation Areas, out of the Everglades’ landscape to provide water for urban areas and agriculture, and to reverse excessive drainage caused by earlier water-control projects. These areas — along with two major highways, Alligator Alley (I-75) and Tamiami Trail (US-41) — divided the Everglades into smaller segments, which inhibited the natural flow of water.
Agriculture affected the Everglades in other ways too. Farmers in the Everglades Agricultural Area cultivated their crops on peat soils. To allow crops to flourish, however, farmers drained the peat soil via canals. Once exposed to air, the drained peat soils were gradually oxidized away by aerobic bacteria that degraded the aquatic plant remnants in the peat soil. This process of peat soil oxidization continues today, and areas that once contained nearly three meters of productive peat soil now have just one-third as much. By 2050, scientists estimate only about 20 centimeters of productive soil will remain. In addition to draining soils, farmers also fertilized the land. Thus, over the years, a variety of agricultural chemicals has been added to the Everglades Agricultural Area’s soil.
Today, the future of the Everglades is uncertain. The development of agriculture, the resulting soil subsidence and the use of agricultural chemicals have had profound impacts on the water quality of the Everglades. Now scientists are working with local, state and federal officials to better understand how society’s pollution has affected the Everglades’ water in an effort to learn how to restore and protect this unique ecosystem.
It wasn’t until 1947, when Marjory Stoneham Douglas published “The Everglades: River of Grass,” that people began to recognize that the Everglades ecosystem was in trouble. The book chronicled the decline of the large population of wading birds that once inhabited the region, and the arrival of invasive plants like the ferns that cover the tree islands and animals that threaten native species.
In the mid-1980s, scientists uncovered other problems. They discovered eutrophication — the overgrowth of plant and algal species due to excess nutrients — in the normally low-nutrient ecosystem was harming the Everglades’ vegetation. The extra nutrients were allowing nutrient-loving plants like cattails to invade the wetlands and displace the sawgrass and other native plant species. Scientists linked phosphorus to the eutrophication problem, and phosphorus quickly became the scapegoat for all of the Everglades’ troubles with water quality.
The phosphorus was entering the Everglades from canal-water discharge originating within Lake Okeechobee and the Everglades Agricultural Area, where farmers enriched their fields with phosphorus-heavy fertilizers. In 1988, the federal government sued the state of Florida over the phosphorus contamination. In 1988 and 1992, Florida settled with the federal government and agreed to take steps to reduce phosphorus contamination in the Everglades. Agricultural interests agreed to limit the use of phosphorus fertilizer, and the state established artificial wetlands — called Stormwater Treatment Areas — on former agricultural land within the Everglades Agricultural Area to help clean the phosphorus-laden canal water before it emptied into the Everglades.
These approaches to restoration resulted in a dramatic reduction in the amount of phosphorus in canal water that enters the Everglades, from about 150 parts per billion in the early 1990s to less than 30 ppb today. Achieving the pre-development levels of 10 ppb may be difficult. Nevertheless, a significant reduction has been achieved.
It was not recognized when these efforts were implemented, however, that phosphorus was not the only chemical contaminating the Everglades and threatening the ecosystem’s water quality. Restoring the Everglades would prove to be far more complex.
During the 1980s and 1990s, there were sporadic reports of high levels of mercury in fish caught in the Everglades. Further study supported by the Florida Department of Environmental Protection revealed an extensive mercury problem in fish throughout the Everglades. Methylmercury accumulates most in wildlife and is the most toxic. In some surveys, levels of methylmercury in fish exceeded 4 parts per million, some of the highest levels recorded in the United States. The public, especially pregnant women, was warned to limit their consumption of fish from the Everglades and Florida Bay. The high levels of methylmercury also presented a risk to fish-eating wildlife, especially wading birds.
Unraveling the complexities of the methylmercury issue in the Everglades has been a challenge. Methylmercury is principally produced by sulfate-reducing bacteria. Inorganic mercury enters the ecosystem through rainfall, and then bacteria convert the inorganic mercury into methylmercury during metabolism. The waterlogged, low-oxygen peat soils of the vast Everglades wetlands are an excellent place for the bacteria to live. And they have plenty of mercury to satisfy their needs. Scientists have discovered that south Florida receives some of the highest levels of mercury deposition in the United States, mostly in rainfall. The mercury deposited on the Everglades appears to travel long distances to get there, probably from worldwide sources: Despite a 90 percent reduction in anthropogenic emissions of mercury from nearby urban areas, levels of mercury deposited in the Everglades have not declined perceptibly.
The extensive wetland area and high levels of mercury deposition help to explain the high levels of methylmercury in the Everglades’ wildlife, but this is not the whole story.
The water quality issues that are degrading the Everglades are the result of land-use decisions — such as establishing a major agricultural area upstream of a large freshwater ecosystem — made in the early to mid-1900s. However, these problems do not necessarily doom the Everglades, provided that we institute the right restoration efforts.
Sulfate contamination, for example, may be improved if officials redesign the Stormwater Treatment Areas, which remove phosphorus from canal water before it enters the Everglades. Although Stormwater Treatment Areas have successfully reduced phosphorus levels, they are much less effective at removing sulfate. They remove no more than 20 percent of sulfate in canal water. A reevaluation of the design of Stormwater Treatment Areas might lead to improved sulfate removal processes.
Lowering the amount of sulfate that farmers put on their agricultural fields, just as they have done with phosphorus, would also help. But at the moment, scientists do not know how much sulfate originates from natural sulfur and how much sulfur is locked in the soil due to past agricultural practices. Soil oxidation may be releasing this sulfur, allowing it to run off into the canals that discharge into the Everglades.
One of the principal goals of restoration is diverting additional water into areas, such as Everglades National Park, that receive insufficient flow. One way to do this is to direct water from the canal network to the water-impoverished areas. But such a move is likely to lead to extensive contamination, because canals have high levels of sulfate. Indeed, some evidence from recent USGS field studies shows that sulfate-laden canal water entering Everglades National Park may be triggering methylmercury production and bioaccumulation there. A better approach would be to slowly flood expansive areas of marshland where sulfate levels can be diluted by the vast amount of water, and microbial sulfate reduction can remove sulfate from the surface water and sequester it as metal sulfides and organic sulfides in soils.
Perhaps one of the most promising restoration developments in the last year was the state of Florida’s proposal to buy 73,250 hectares of land in the Everglades Agricultural Area from the U.S. Sugar Corporation at a cost of $1.34 billion. The purchase would end agricultural practices, including the use of fertilizers, on approximately one-third of the Everglades Agricultural Area’s fields. Although not a panacea for restoring the Everglades, this plan should help reduce phosphorus and sulfate loads — and as a result, also reduce methylmercury production. If the purchased property is kept flooded, it will also reduce sulfate fluxes to the Everglades by alleviating soil oxidation and keeping sulfur firmly sequestered in reduced form in the soil. And someday, the acquired Everglades Agricultural Area land could eventually return to a functioning wetland, swamp or marsh.
Poor water quality in wetland ecosystems is rarely caused by a single culprit. In hindsight, the initial restoration focus on phosphorus alone was somewhat unfortunate. Phosphorus remains an important water quality issue, but understanding how sulfate, mercury and other contaminants affect the Everglades and devising innovative mitigation strategies are also critical for a successful restoration of this ecosystem. This requires understanding the sources, biogeochemical cycling and ecosystem sinks for various contaminants, as well as examining ecologic sensitivities to these contaminants. In the short run, this may require trade-offs in balancing water quantity needs with water quality challenges, because much of the available water contains contaminants. But by working together, state, federal, university and private-industry researchers are trying to bring back the good health that the Everglades’ plants and animals once enjoyed, and restore this unique ecosystem for future generations to appreciate.
© 2008-2021. All rights reserved. Any copying, redistribution or retransmission of any of the contents of this service without the expressed written permission of the American Geosciences Institute is expressly prohibited. Click here for all copyright requests.